Our client Emotra develops device that helps fight against suicide risk among teenagers
Now kids are back to school we as parents take notice of how they develop in that environment. Some of them may suffer from social pressure or even some sort of violence that can lead to depression or even worse. The 10th of September is the World Suicide Prevention Day and we at Inspiralia are proud of having worked with clients like Emotra to fight against this problem.
Emotra is a Swedish company founded in 2001 with the objective of bringing to the market the result of our many years of R&D on the field of suicide risk detection and prevention: EDOR®-BOX.
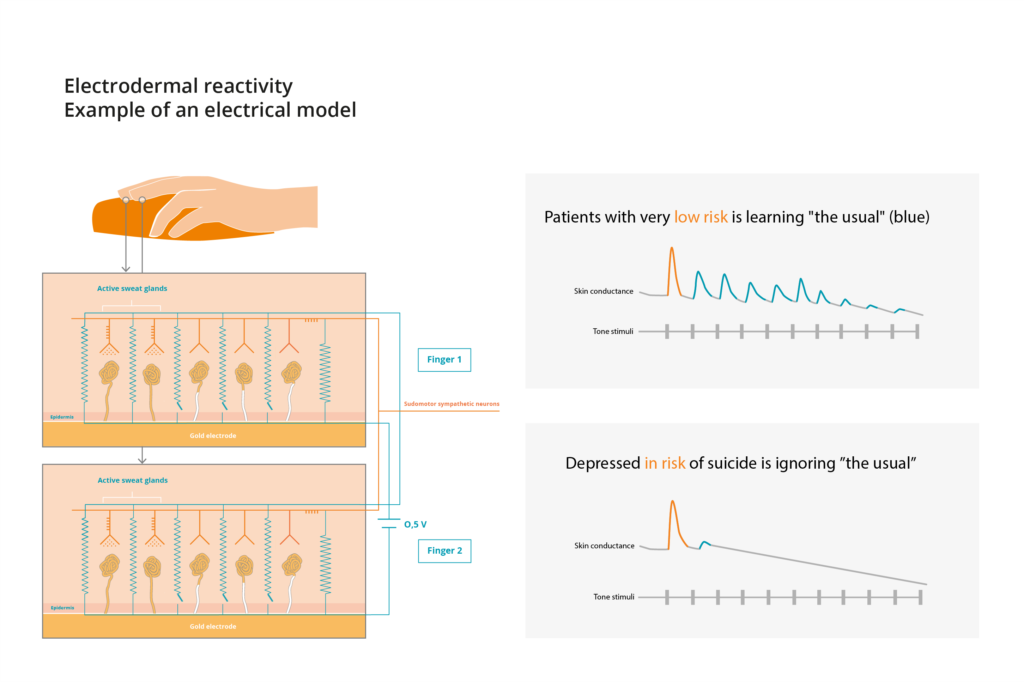
EDOR®-BOX is a CE-Marked medical device that measures electrodermal activity in the patient’s fingertips, to analyze reaction patterns to sound, in patients that show signs of depression. Abnormal reaction patterns are identified as hyporeactive, meaning significantly reduced response to sound stimuli, and are related to high risk of suicide and suicide attempts, functioning as a biomarker for high-risk patients.
EDOR®-BOX encloses a cost-effective, quantitative, objective and non-invasive biomarker-based solution to facilitate the diagnosis and early detection of patients undergoing a propensity to attempt suicide. EDOR®-BOX is also the first of its kind device for prediction of suicide propensity, as no similar solution exists on the market or in development.
The test takes only 30 minutes to be conducted and documented, being completely user-friendly for the healthcare professionals, since it needs less than 4 hours of training.
The system has been validated in over 1500 adult patients of 16 clinics, in the multicentre EUDOR-A study, proving satisfactory results.